China Drugmakers Climb the Value Chain as a National 'Pillar Industry'
Reading into China's 15th Five-Year Plan
China's 15th Five-Year Plan (2026-2030) will be adopted at the ongoing Two Sessions. The plan outlines how the nation will move to achieve basic modernization by 2035, amidst a more disquieting global environment as well as China's efforts to become more self-reliant in technology and improving living standards. This series provides insights into the nation's vision in various sectors.

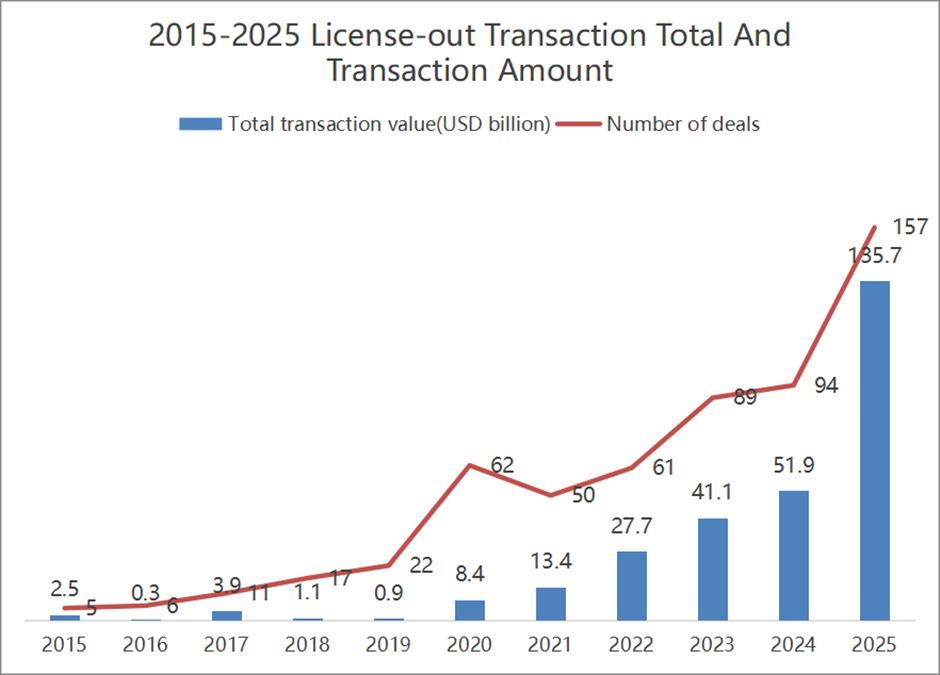
China's innovative drugmakers are off to a blistering start this year, with biotech companies already having struck 44 licensing deals with global drugmakers valued at up to US$53.3 billion – more than a third of last year's total.
Recent transactions include Remegen's US$5.6 billion partnership with AbbVie and a potential US$8.85 billion collaboration between Innovent Biologics and Eli Lilly.
The momentum comes as Beijing signals stronger support for the biotech sector. In his report to the National People's Congress last week, Premier Li Qiang said China will work to develop biotechnology and pharmaceuticals into new "pillar industries."
Data from industry tracker Pharmcube shows that Chinese companies signed 186 overseas licensing deals valued at US$137.7 billion in 2025, nearly 10 times the level seen in 2021.
The surge in dealmaking is backed by a rapidly expanding research pipeline. According to Citeline's Pharmaprojects database, China had more than 7,000 drugs in development in 2025, accounting for over 30 percent of the global pipeline and ranking second worldwide.
"More Chinese drug candidates are now being evaluated within multinational pharma companies' core pipeline strategies," said Wu Xiaoying, co-leader of Ernst & Young health science and wellness sector. "That suggests Chinese assets are moving further up the global value chain."
At the same time, the structure of Chinese drugmakers is changing.
"We are seeing more diversified partnership structures emerging," Wu said. "Chinese companies are increasingly willing to retain part of the drug rights, rather than exiting entirely at an early stage."
In the past, many Chinese biotech firms licensed out overseas rights at an early stage of development, a model often described in the industry as "selling seedlings."

Recent transactions reflect that shift.
In February, Innovent Biologics expanded its partnership with long-time collaborator Eli Lilly to jointly develop new molecules in oncology and immunology. Under the agreement, Innovent will lead research from drug discovery to proof-of-concept studies in China, while Lilly will hold global rights outside China. The deal includes US$350 million in upfront payments and up to US$8.5 billion in potential milestone payments.
Other partnerships have introduced more complex financial structures. Harbour BioMed, for example, agreed late last month to license its antibody therapy HBM4003 to Solstice Oncology outside China in a transaction combining licensing payments with equity investment, with upfront consideration exceeding US$100 million.
Meanwhile, some Chinese drugmakers are licensing out platform technologies rather than individual products. In January, CSPC Pharmaceutical Group signed a research agreement with AstraZeneca to develop long-acting peptide therapies using its drug-delivery and AI drug-discovery platforms.
The trend shows that multinational drugmakers are increasingly willing to share development risks and future profits with Chinese partners – a sign of confidence in China's research capabilities.
"A few years ago, when we did license-out deals, we were basically selling overseas rights outright," said the founder of one Chinese biotech company. "The partner paid an upfront fee, and what happened later had little to do with us. Now multinational pharma companies often want us to keep part of the rights. We share the development risks and the future rewards. That kind of arrangement shows they increasingly see us as real partners in drug development."
But Wu also noted that whether companies can retain more rights ultimately depends on the strength of the asset and the clinical data. "Only projects with strong data and clear differentiation tend to secure deeper collaboration," she said.
Researchers Sun Yin and Wu Wei from Zhejiang University estimate that Chinese companies often capture only about 5 to 8 percent of value in typical licensing deals, despite taking on much of the early-stage development risk.
In one widely cited case, a Chinese biotech firm licensed out a bispecific antibody candidate for an upfront payment of US$25 million in 2023. When the asset was resold last year, the upfront payment alone had jumped to US$1.5 billion.
"Chinese companies are gradually gaining more room in negotiations, but the shift is happening through deal structures rather than a sudden jump in profit share," Wu said. "Ultimately, bargaining power comes down to how differentiated the asset is, how strong the clinical data are and whether the company can participate in global development. As those capabilities improve, Chinese companies will naturally gain more negotiating space in future deals."
Recently, Chinese biotech firm ImmuneOnco and US-based Instil Bio said they had terminated a licensing agreement signed in August 2024, with global rights to two cancer drug candidates reverting to ImmuneOnco. The move marked the first high-profile "return" of an overseas licensing deal for China's innovative drug sector this year.
An executive at a Chinese biotech company who previously experienced such a terminated licensing agreement said financial strength and execution capability of overseas partners can sometimes matter more than the size of the deal.
"A few years ago, we were too focused on the partner's big-name reputation," he said. "We didn't fully assess their cash flow or pipeline priorities. Once they decide to scale back their pipeline, our product can quickly become expendable."
Wu said such risks are particularly high for projects lacking clear differentiation.
"Many companies have concentrated their research on a few popular targets, such as PD-1-related therapies or GLP-1 metabolic drugs," she said. "When multinational pharma companies screen potential partnerships today, what they value most is differentiation and clinical value. If a project is simply another version of an existing therapy, even if the technology is mature, the room for partnership is often limited."
Investment bank UBS forecasts that China-developed innovative drugs will grow at an annual rate of about 20 percent over the next five years, with their share of China's pharmaceutical market rising to 59 percent by 2030.
Morgan Stanley projects an even larger global footprint. By 2040, Chinese-origin drug assets could account for about 35 percent of new drug approvals by the US Food and Drug Administration, up from just 5 percent in 2024, while annual overseas revenue from Chinese innovative medicines could reach US$220 billion.
"China is likely to evolve into one of several major innovation contributors alongside the US and Europe," Wu said.
She added that Chinese companies are likely to participate in global drug development through a more diverse set of models. However, moving up the pharmaceutical value chain will require more than dealmaking.
"One immediate gap is the global acceptability of clinical data," Wu said. "It is not only about running trials properly, but also about study design, endpoints, statistical methods and communication with international regulators."
Commercialization capability, particularly in overseas markets, will also become increasingly important, she added.
"Ultimately, the question is whether innovation can be turned into globally recognized products that succeed in the marketplace," she said.
Editor: Liu Qi




