China Overhauls Drug-Pricing System to Allow Wider Market-Driven Forces
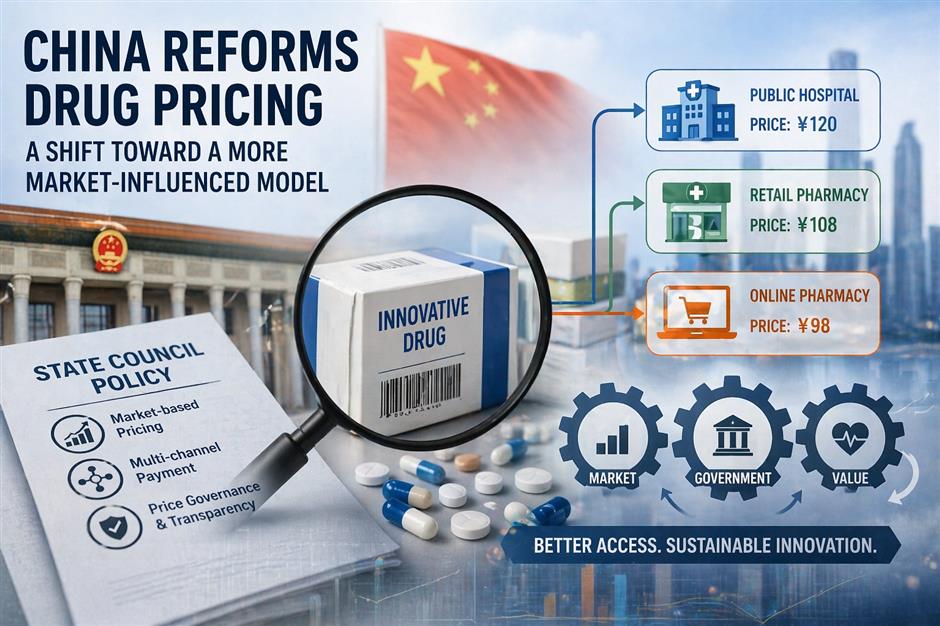
A patient searching for the same drug in China might find three different prices: one at a public hospital, another at a neighborhood pharmacy and a third on an e-commerce platform.
That discrepancy is no longer just a quirk of China's fragmented healthcare system. It is becoming part of a broader policy shift.
China has begun overhauling how drug prices are set, moving away from a system dominated by state-led negotiations toward a more market-driven model that could reshape how innovative medicines are priced, marketed and ultimately valued.
Under a new policy released last week by the State Council, China's Cabinet, pharmaceutical companies will be given greater flexibility to set initial prices for newly launched drugs, particularly advanced therapies, while state-backed payers such as the National Reimbursement Drug List will continue to play a central role in determining long-term affordability.
The change addresses a long-standing tension in China's pharmaceutical sector – where drugmakers undertaking costly research and development find it hard to recoup costs when products that hit the market are constrained by price controls.
"The policy combines an effective market with a well-functioning government supporting what should be supported, guaranteeing what should be guaranteed and regulating what should be regulated," Shi Zihai, deputy commissioner of the National Healthcare Security Administration, said at a State Council press briefing.
For years, the commercial path for innovative drugs was relatively fixed: launch, enter the government reimbursement program and accept steep price cuts often exceeding 70 percent. That dynamic has been widely cited as one factor behind the steady wave of deals where Chinese biotech companies seek profits through licensing agreements with global pharmaceutical groups.
The new framework suggests policymakers are attempting to rebalance that equation.
"It is a positive signal that helps bring pricing strategies back to the fundamental value of innovation," Akeso, a Chinese biopharmaceutical company developing innovative biological medicines, told China Biz Buzz.
"In the past, the market often focused excessively on price itself," he went on to explain. "The new policy emphasizes market-based pricing, meaning payers and regulators are placing greater recognition on differentiated clinical value."
Akeso pointed to its two marketed, bispecific antibodies as examples, saying their clinical performance, supported by both trial data and real-world evidence, could justify pricing that better reflects their therapeutic value, at least in the early stages of commercialization.
"Pricing is no longer solely the outcome of reimbursement negotiation, but increasingly incorporates the market's assessment of clinical value," the company added.
The policy changes are not limited to how drugs are priced at launch or reimbursed over time. They also extend to how prices are formed and adjusted across different distribution channels – an area that has long been a source of variation in China's healthcare system.
Public hospitals remain the primary sales channel for drugs, while retail pharmacies and online platforms have expanded rapidly in recent years.
"These channels operate under different market conditions and pricing mechanisms, which explains why the same drug may be sold at different prices," Wang Xiaoning, director-general of the Department of Drug Pricing and Procurement, told the briefing.
He noted such variations are influenced by factors including business models, procurement channels, supply chains and operational efficiency.
"Price differences are normal, but they should remain within a reasonable range," he said, adding that narrowing excessive gaps across channels has become a key focus of regulatory efforts. "Under competitive pressure and improved governance, prices across different channels are gradually converging and becoming more rational."
Beyond narrowing price gaps, policymakers are also reshaping how drugs are ultimately paid for, a shift that could have a more profound impact on the industry's commercial model.
Alongside the national public insurance system, authorities are encouraging the expansion of multi-channel payment mechanisms, including commercial insurance and out-of-pocket markets, which could gradually reduce the industry's heavy reliance on reimbursement program inclusion.
"National Reimbursement Drug List inclusion will remain the main pathway for much patient access in the near term," Akeso said. "But a multi-layered payment system will bring more flexibility and segmentation."
This evolving structure places China somewhere between the world's two dominant pricing models.
In the US, pharmaceutical companies retain significant control over launch prices, with insurers and intermediaries negotiating discounts afterward. In Europe, governments typically anchor prices to clinical value through formal health-technology assessments, exerting stronger upfront control.
China's emerging approach blends elements of both, allowing greater pricing autonomy at launch while maintaining regulatory oversight through reimbursement, procurement and price monitoring mechanisms.
The result is neither a fully market-driven system nor a purely state-controlled one, but what industry participants increasingly describe as a "negotiated market."
At the same time, authorities are rolling out nationwide drug-price comparison tools, allowing patients to check real-time prices across pharmacies, a move designed to use transparency and consumer choice to discipline pricing.
For the industry, the policy signals both opportunity and constraint.
On one hand, greater flexibility in early-stage pricing and diversified payment channels could improve returns for companies developing highly innovative therapies, particularly those targeting unmet clinical needs.
On the other, the government has made clear that it will continue to curb irrational price competition, especially in procurement programs where aggressive bidding has, in some cases, raised concerns about long-term supply stability and innovation incentives.
"The policy emphasis on preventing irrational price competition is a positive signal, encouraging the industry to return to value-based innovation," Akeso said. "Pricing pressure has emerged in certain crowded therapeutic areas. When multiple products fail to demonstrate differentiated clinical advantages, price naturally becomes the main competitive tool."
China's once low-price market shaped by state negotiations is indeed becoming a more complex commercial environment, where pricing strategy, channel management and payer mix will play an increasingly decisive role.
"The policy reflects a growing regulatory focus on steering the industry back to value-based fundamentals," Akeso said. "For companies built on differentiated, clinically driven innovation, it is a clear incentive."
Editor: Yao Minji
In Case You Missed It...








