CorrectSequence Therapeutics Signals a Safer Path for Gene Editing with Base Editing Breakthrough

Gene editing has delivered a series of scientific breakthroughs over the past decade. Turning those breakthroughs into viable businesses has proved far more difficult.
The latest early clinical results are notable not for introducing a new drug candidate but for suggesting a change in gene editing technology development.
A study in Nature found that CS-101, a therapy developed by CorrectSequence Therapeutics in Shanghai and its academic partners, reduced the need for blood transfusions in patients with β-thalassemia within weeks.
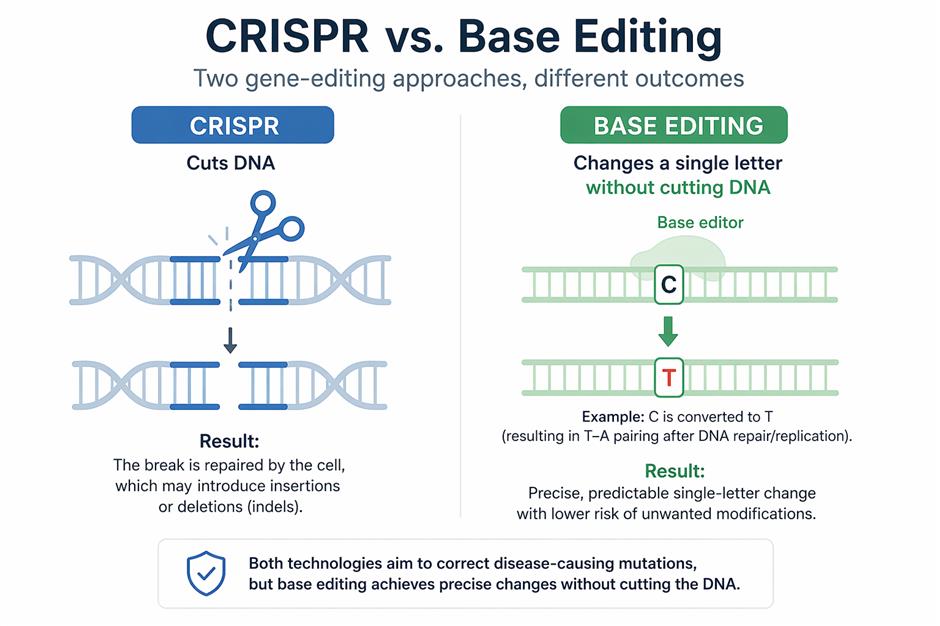
What sets CS-101 apart is not the disease it targets but the editing method behind it. Traditional CRISPR-based therapies cut DNA strands, but base editing changes individual genetic letters without breaking the DNA, which helps lower the risks linked to cutting DNA.
"Our proprietary developed base-editing system is a foundational platform technology that can be readily adapted to a wide range of indications, significantly reducing both costs and manufacturing complexity," said Mou Xiaodun, CEO of CorrectSequence Therapeutics.
For years, gene editing has been dominated by CRISPR-based approaches. Therapies created by companies like Vertex Pharmaceuticals and CRISPR Therapeutics are already available for some conditions, showing positive results but also revealing issues like high costs, complicated production, and difficulty in making them on a larger scale.
Parallel to these developments, companies are exploring alternative approaches. One of the first base editing companies, US-based Beam Therapeutics, is developing its pipeline for clinical use as a more precise gene modification method.
The emergence of CS-101 adds a new data point to that landscape.
Early data suggests speed and durability advantages. Patients in the CS-101 study achieved transfusion independence within 16 days of treatment and maintained hemoglobin levels for over a year. The therapy has been applied to nearly 20 patients with β-thalassemia and sickle cell disease.
CS-101 entered clinical testing in October 2023, becoming the world's first ex vivo base-editing therapy to reach the clinic. The first patient has remained transfusion-independent for more than 28 months.
Most gene-editing therapies for blood disorders use an ex vivo model, where a patient's cells are extracted, modified in a lab, and reinfused. Similar to CAR-T therapies, the process requires customized manufacturing and infrastructure, which has limited industry adoption.
"From a therapeutic strategy perspective, in vivo gene editing offers greater versatility and lower cost," said Chen Jia, co-founder of CorrectSequence Therapeutics and director and professor of the Gene Editing Center at ShanghaiTech University. "Our strategy is to expand base editing across both in vivo and ex vivo settings and to move from rare diseases toward more common conditions."
The challenge is not unique to one company. Across the field, gene-editing technologies continue to face the same fundamental question: whether strong clinical outcomes can translate into scalable, economically viable treatments.
That experimentation is expanding in China. Companies like CorrectSequence Therapeutics are advancing early-stage programs with faster patient recruitment and a growing clinical research ecosystem to generate clinical data faster than in other markets.
"Translating early clinical success into a scalable business model requires manufacturing scale-up, quality validation and clinical trials. We are advancing clinical development and the regulatory pathway toward commercialization," Mou said.
For now, base editing remains an emerging approach rather than an established platform. But as more clinical data accumulates, the competitive landscape of gene editing may increasingly be shaped not just by scientific breakthroughs but also by which technologies can be translated into sustainable business models.
Editor: Yao Minji




